Technology & Products
[Battery Glossary] Separator, Electrolyte, SEI Layer
2025.12.02
|
[Battery Glossary] answers the questions related to batteries with key term explanation. From fundamental battery principles, manufacturing processes to emerging next-generation technologies, [Battery Glossary] makes battery concepts easy to understand. |
Separator

A separator serves as a physical barrier between the cathode and anode. This is crucial because direct contact between the cathode and anode can lead to a short circuit, potentially causing fire or explosion. Separators are typically made from polymer materials such as polypropylene (PP) and polyethylene (PE).
The surface of the separator contains pores that are invisible to the naked eye, allowing lithium-ions move between the cathode and anode. Separators are generally classified into wet or dry types depending on how these pores are formed. Dry separators create pores by pulling PE and PP membranes during solidification. While this manufacturing process is relatively simple, it results in inconsistent pore sizes. In contrast, wet separators are produced by extracting films after adding solvents and additives to polymer materials. As the film cools, the solvent is extracted, creating uniform pores. However, this process is more complex and therefore more expensive.
Electrolyte
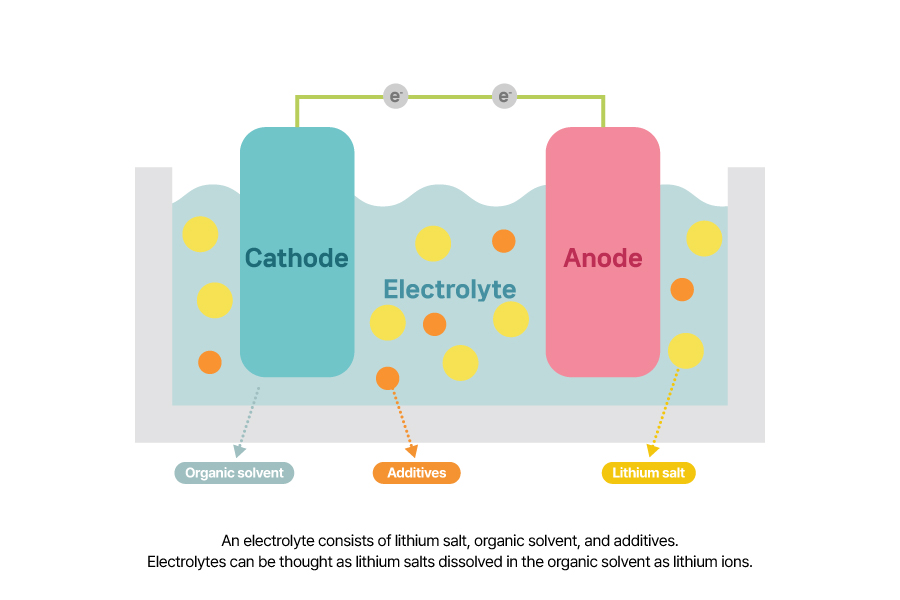
An electrolyte is a substance that allows lithium ions to travel between the cathode and anode freely. It typically consists of a lithium salt, organic solvent, and additives. You can think of it as lithium salt dissolved in an organic solvent as lithium ions.
When lithium ions leave the cathode, they push the dissolved lithium ions in the electrolyte forward. Through this chain movement, the lithium closest to the anode, finally enters the graphite layers. Therefore, using an electrolyte with a high concentration of dissolved lithium ions enhances ionic conductivity, which is why it’s crucial to select lithium salts that readily dissociate into lithium ions.
SEI (Solid Electrolyte Interphase) Layer
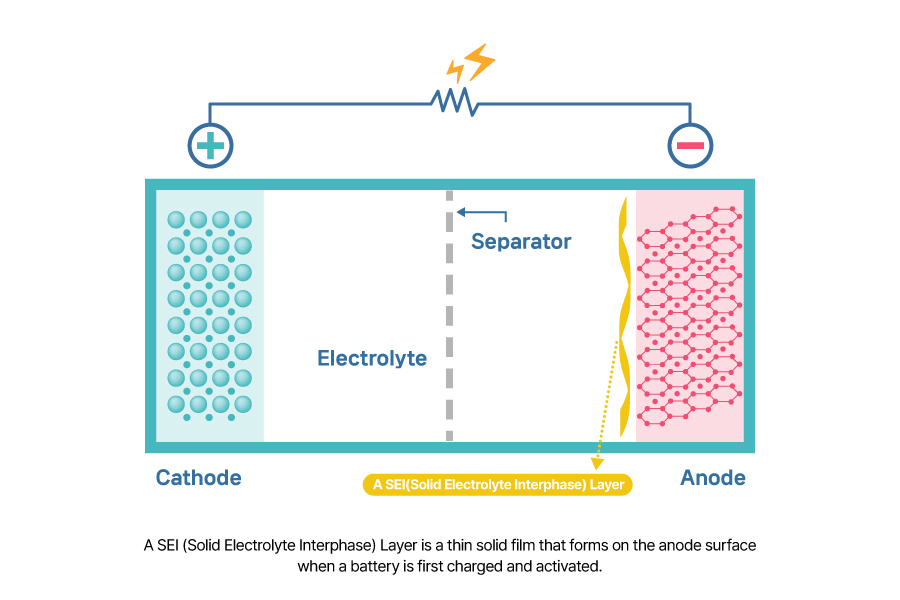
A SEI (Solid Electrolyte Interphase) Layer is a thin solid film that forms on the anode surface when a battery is first charged and activated. During the initial electrochemical reaction at the anode surface, the electrolyte decomposes, generating a thin layer of solid electrolyte.
The SEI layer plays a critical role in battery performance and lifespan. Once formed, it prevents further electrolyte decomposition and lithium consumption which can help preserve the battery’s capacity. Additionally, since only lithium ions can pass through the SEI layer, it helps maintain the structural stability of graphite during charge and discharge cycle.
