Technology & Products
[Battery Glossary] Anode Active Material, Graphite, Silicon
2025.10.16
|
[Battery Glossary] answers the questions related to batteries with key term explanation. From fundamental battery principles, manufacturing processes to emerging next-generation technologies, [Battery Glossary] makes battery concepts easy to understand. |
Anode Active Material
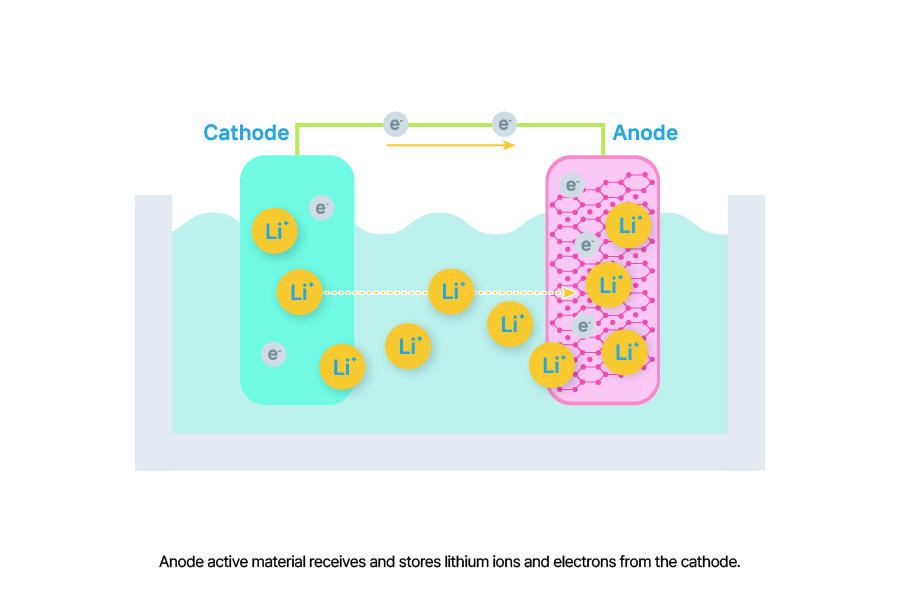
Anode Active Material refers to a material that receives and stores lithium ions and electrons from the cathode during battery charging, and releases the stored lithium ions and electrons back to the cathode during discharging. During this process, electrons move through an external circuit, allowing current to flow.
Similar to cathode active materials, anode active materials significantly influence a battery’s capacity. Even if a cathode active material can hold a large number of lithium ions and electrons, the battery cannot perform effectively if the anode active material cannot receive or store them. Typically, graphite is used as the anode active material in lithium-ion batteries, sometimes mixed with small amounts of silicon.
Graphite
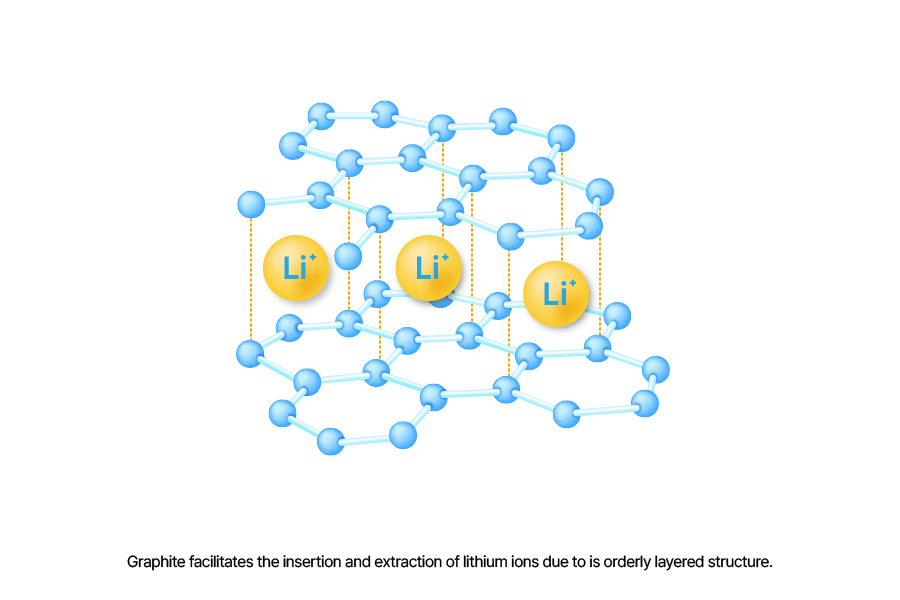
Graphite is a material used as the anode active material in lithium-ion batteries. Its orderly layered structure facilitates the insertion and extraction of lithium ions during charging and discharging.
Depending on the production method, graphite is classified into natural and synthetic types. Natural graphite is mined from nature, so it is readily available and inexpensive. Synthetic graphite is produced by heating natural graphite at temperatures above 3,000 degree Celsius. Due to its lengthy and complex production process, synthetic graphite is more costly, which leads to mixing with natural graphite.
Graphite directly affects battery lifespan. During charging, lithium ions move to the anode and are stored between graphite layers, causing the graphite’s volume to expand and contract by about 10%. While this is a natural occurrence, it can degrade the graphite’s structure in the long term, impacting battery lifespan.
Silicon

Silicon is considered a potential successor to graphite as an anode active material in lithium-ion batteries. Its advantage lies in its higher capacity to store lithium ions compared to graphite. This higher capacity allows for thinner anode plates, making rapid charging more efficient.
However, silicon has a notable drawback: large volume expansion. During the insertion and extraction of lithium ions, graphite’s volume expands by around 10%, whereas silicon’s volume can expand up to 400%. This is detrimental to the battery, so only small amounts of silicon are currently mixed into anode active materials.
