Technology & Products
[Battery101] Anode Materials Determine the Safety and Lifespan of Li-ion Batteries
2024.07.05
|
101[wʌ́nouwʌ́n] means basic knowledge of a topic or collection of introductory materials to a topic. Our Battery 101 series talks about all things battery: the history, technical aspects (basic principles and mechanisms), industrial aspects (IT, electric vehicles, ESS, etc.), and next-generation technologies that SAMSUNG SDI will innovate while opening up its future. Batteries have infinite potentials that exceeds our wildest imagination. Through Batteries 101 series, you will have a chance to see the entire spectrum of the battery's possibilities and to conjure SAMSUNG SDI’s pivotal role in it. |
The quantity and quality of graphite affect the capacity
The cathode substrate (aluminum) of battery sends electrons from cathode to anode when the power is connected for charging. When this happens, electrons move through tabs—the poles of the battery—as they cannot pass through the electrolyte. Lithium ions, however, pass through the electrolyte and flow toward the anode, unlike electrons. As such, both electrons and lithium ions move to the anode.
Then, where do the lithium ions and electrons stay after they reach the anode?
Graphite used as an anode active material is composed of graphene layers—multiple layers of carbon atoms—and lithium ions and electrons settle in this lattice structure of graphite just like residents moving into an apartment floor by floor. The battery charging is completed when most of the lithium ions have traveled to the graphene layers.
Strictly speaking, the electrons in those graphite layer are not in any way a stable state. Electrons released from lithium and lithium ions tend to return to lithium metal oxide cathodes. Because of this tendency, during discharging, electrons and lithium ions move towards the cathode like a ball sliding down a hill whereas graphite anodes only serve to store lithium ions. In other words, graphite, the main anode material, does not affect the chemical reactions of lithium-ion batteries.
Graphite is the most popular choice for an anode material because of its high conductivity of electrons and robust intercalation of lithium ions. Anode materials play a crucial role. Both quantity and quality of graphite are directly linked to increasing the battery capacity. Even if you add a large volume of active materials to the cathode to send electrons, this would not affect the performance of a battery if the anode fails to take in such flows of electrons and lithium ions.
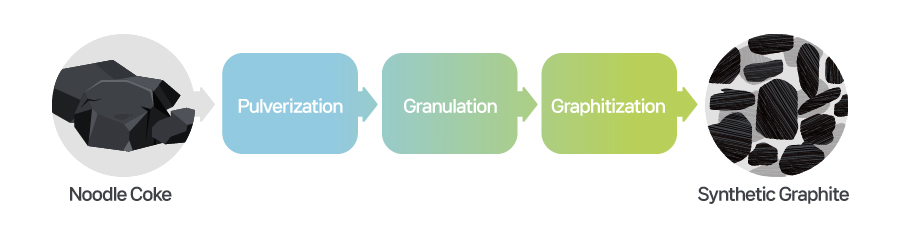
[Manufacturing process of synthetic graphite and crystallized images of natural graphite and synthetic graphite]
Graphite, the primary anode material for lithium-ion batteries, is largely classified into natural graphite and synthetic graphite. Although both natural graphite and synthetic graphite are used in the market, the share of synthetic graphite is gradually increasing these days. Hard carbon used an anode material firstly was a carbon-based material (a type of low-crystallinity carbon) but has not been in use for long due to its small capacity, despite having the highest output.
Natural graphite is an underground resource that is formed during metamorphism of carbon compounds in the sediments under high temperatures and pressures for an extended period of time. Due to this, natural graphite consists of graphene layers, in which lithium ions travel through the lattice structure. Since natural graphite has limited ion transport paths, it comes with disadvantages like low charge & discharge efficiency. It is also more prone to swelling as layers in graphite expand while ions move back and forth during charging and discharging. Nevertheless, natural graphite is still an attractive option as it is less pricy than synthetic graphite and the initial charge efficiency has increased with the advance of surface treatment technologies.
Synthetic graphite, on the other hand, is produced by applying heat to coke and pitch at high temperatures over 3,000°C. It has longer a lifespan and higher outputs compared to natural graphite, because artificial graphite has many paths for the movement of lithium ions. This structure increases efficiency and enables fast charging capability in batteries. Moreover, synthetic graphite has a stable isotropic structure, resulting in less swelling and a relatively longer battery lifespan. Owing to its longer and more complex production process, however, synthetic graphite is more expensive than natural graphite.
Research on anode materials continues
Lithium ions are placed inside the graphite's layers when they migrate from cathode to anode during charging. This is also when the battery volume repeats expanding and shrinking by about 10%. It is a natural course in using batteries but in the long term, this swelling is detrimental to battery's performance. As the battery volume constantly changes during charging and discharging, slight changes occur inside the battery as well. For instance, the structure of graphite may collapse or gas may form, which is quite dangerous. In the end, such changes affect the lifespan of the battery.

[Characteristics of anode materials]
As such, there are ongoing research efforts on anode materials so as to improve the safety and performance of batteries. These days, silicon is mentioned as a potential substitute for graphite for lithium-ion batteries. Silicon has higher capacity per weight and enables faster charging and discharging than graphite. More than anything, it can store lithium ions at a much higher rate than graphite can. While graphite stores up to one lithium ion for every six atoms (LiC6) on average, silicon takes up to 22 lithium ions for every 5 atoms (Li4.4S). Therefore, it is possible to produce high-capacity batteries by using silicon as the anode active material and adding more cathode materials.
However, the biggest problem lies in the high expansion rate of silicon. The volume of graphite expands by around 10%, but that of silicon does so by up to 400%. In addition, the destruction of solid electrolyte interphase (SEI) is also an issue. This thin protective layer is formed when the electrolyte reacts with the active materials. It protects anode active materials from the electrolyte while working as a transport path of ions. Using silicon as an anode active material requires caution as this thin layer may be damaged with the expansion of silicon.
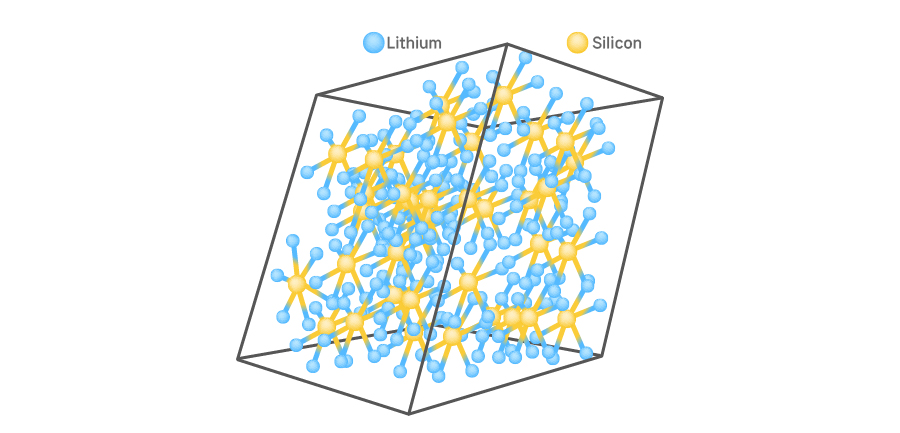
[Lithium ions stored in silicon anode material]
Accordingly, researchers are focusing on maximizing the benefits of silicon and solving issues associated with the expansion of silicon. To this end, there are approaches to make smaller silicon particles or design carbon coating of silicon nanoparticles. SAMSUNG SDI developed and patented Si-carbon nanocomposite (SCN) material. SCN is a material that integrates graphite with nano-sized silicon, which is equivalent to one-thousandths of the thickness of a single strand of human hair. The idea here is to bring together the best attributes of silicon and graphite. These compounds also have a high energy density while addressing the volume expansion issue.
