Technology & Products
[Battery101] Chemistry of Battery
2024.06.07
|
101[wʌ́nouwʌ́n] means basic knowledge of a topic or collection of introductory materials to a topic. Our Battery 101 series talks about all things battery: the history, technical aspects (basic principles and mechanisms), industrial aspects (IT, electric vehicles, ESS, etc.), and next-generation technologies that SAMSUNG SDI will innovate while opening up its future. Batteries have infinite potentials that exceeds our wildest imagination. Through Batteries 101 series, you will have a chance to see the entire spectrum of the battery's possibilities and to conjure SAMSUNG SDI’s pivotal role in it. |
Ionization energy generates electricity
A battery is a device that generates electricity based on chemical energy. That’s why you need to understand chemistry to delve into the world of batteries. The first to look at is ‘ionization energy.’
So we talked about Voltaic pile in one of the previous chapters. Alessandro Volta made a structure consisting of silver (or copper) and zinc discs with salt-water soaked pieces of paper between them. He took it further by placing two different metals in an acid solution and confirmed the electric current was produced when the exposed ends of those metals were connected. That’s how the first-ever chemical cell – Voltaic pile – was invented. How do you know what metals and what acidic solutions to use here? Why did Volta go for silver (or copper) and zinc discs in his Voltaic cells?’ The answer lies in ionization energy.
Elements vary in their respective reactivity to water or air. For example, iron (Fe) easily gets rust in the air or water whereas gold (Au) doesn’t seem to go through any changes no matter where it is. It is because they all have different atomic structures. The easier an element loses an electron, the higher ionization energy it has, meaning it will react to water or air much more easily.
Look at the chart of elements in the order of their ionization energy in the below. You see lithium tops the list and gold (Au) is at the bottom. Zinc used in Volta cells is located in the middle.
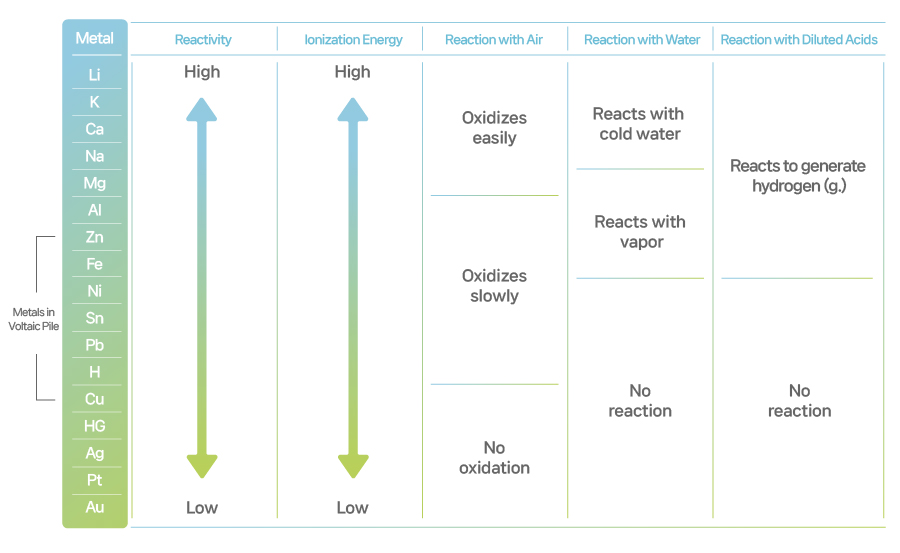
[Ionization Energy of Elements]
We are going back to Volta pile again. Electrolytes are acidic, therefore containing hydrogen (H). According to the chart, zinc has higher ionization energy than hydrogen and a copper (Cu)’s value is lower. That makes zinc soluble in acidic electrolytes whereas copper remains non-reactive. Here, zinc gives out positive ions, leaving electrons only on the zinc electrode. These electrons travel through the wire to the copper electrode, creating electricity. When transferred to the copper electrode, electrons react with hydrogen ions in the acidic solution, resulting in gaseous hydrogen molecule (H2) as proof of that electricity.
The fundamental reason that electrochemical cells can generate electricity is the differing ionization energy levels among the substances serving as anode, cathode, and electrolytes. The wider the difference of those substances in the ionization energy levels, the stronger the electricity generated. Higher ionization energy means creating stronger electricity. These metals rust easily when exposed to the air and reacts explosively when put in water. Lithium, the element with the highest ionization energy, therefore features the most intensive level of reactiveness and that’s why you need extra care and caution in handling the element.
Three units of a battery
People have heard of terminology like volts, watts, and amperes.
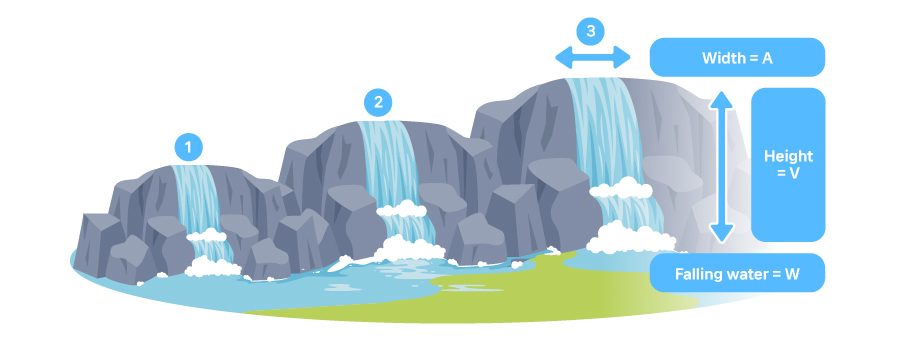
[How power (W), current (A) and voltage (V) work]
In the picture of three waterfalls above, each waterfall has different widths, heights, and the amount of falling water. Number 1 and 2 have the same height, which means that the wider second waterfall carries more power. With the second and third side to side, since the third waterfall is higher, it pushes water with higher power.
Now, let’s take that analogy to batteries. The third waterfall is a battery with the highest voltage of the three. Voltage is electric potential difference between two points. Here, the height of a waterfall indicates voltage. So, the higher the waterfall, the bigger the voltage, therefore, bigger difference of electric potential between cathode and anode. Usually household applications use 220 volts and industrial applications adopt higher 380 volts. Some small digital gadgets have 5, 12, or 19 voltage batteries because they can operate with relatively lower voltage.
Next, amperes (amps). Ampere is the unit of electric current passing through wires, represented in the symbol A (ampere). One ampere is equal to 1 coulomb (electric charge) moving past a point in 1 second.
Then which waterfall has the largest volume of water flowing from top to bottom? That would be the third one. That also means that the third waterfall is a battery that has the biggest power (watts). Power is the amount of electric energy consumed and it is expressed in watt, meaning energy transferred or converted per one second.
A battery’s power is calculated by multiplying the voltage and the current. Take for instance, a battery with 220V and 10A. It has 2,200W power. It has the same mechanism with the volume of water flow in the waterfalls is calculated by the multiplication of per-unit time flow and the amount of time,
Power (W) = Voltage (V) × Current (A)
Taking a step further, let’s find out more about the units used when talking about batteries: ampere-hour (Ah) for electric charge, watt-hour (Wh) for energy, and watt-hour per liter or kilogram (Wh/L or Wh/kg) for energy density. If a waterfall is equivalent to electricity, batteries would make a dam because they store electricity which gets used and refilled again. Therefore, a battery’s capacity represents the total amount of electricity stored in that battery, analogous to the volume of water stored in a dam. A battery-run device’s operating time depends on the battery’s capacity. So the formula of battery capacity comes as follows: the discharge current (A) multiplied by the discharge time (hour), hence an ampere-hour (Ah).
A battery's energy is the total energy held by the stored electricity, which would be equivalent to the total energy held by the stored water in the dam. The energy is proportional to the battery voltage and the discharge current, so is calculated by multiplication of the battery voltage (V) with the current (A) and time (hour). The unit for battery energy is therefore a watt-hour (Wh).
Last but not least, energy density. By energy density, we measure the amount of electric energy stored per volume or weight. That is, in the dam analogy, the amount of energy stored in the water per volume. You calculate a battery's energy density by dividing the electric energy (Wh) by the volume (L) or weight (kg), therefore, using the units like Wh/L or Wh/kg.
What do you do when you want to increase voltage? The answer is in series. Having batteries in series increases the voltage whereas having them in parallel increases the current. When you are charging a battery, having high volts and currents is better because a high volt brings in strong power and a high current lets a lot of electric currents flow.
Standard reduction potential indicates a battery’s power
All battery cells carry their own voltage, such as 1.5V or 3.5V. How can these batteries be built to have certain voltage? The answer is the standard reduction potential.
As said before, Volta chose his elements of a cathode and an anode for his battery based on the ionization energy levels. Metals with higher ionization energy easily lose electrons, being more prone to oxidation. Volta set up a basic framework of batteries that uses the ionization energy of elements but failed to make batteries with high voltage and long running time.
That tells you that simply knowing how the ionization energy works was limiting when you wanted to make a good battery. Even if you could tell one metal had higher ionization energy than another, there was no way finding out just exactly how much that difference was. For that very reason, you couldn’t know how much voltage a battery would have until you measured the battery itself. Without a theoretical value, an experiment becomes a lost ship at sea.
Of course, we don’t have that problem anymore, thankfully, ever since the standard reduction potential (E°) was defined, which specifically calculates the ionization energy of metals.

[Electromotive force of secondary batteries]
In chemical cells, a battery’s voltage generated via redox reactions is an effect of what’s called electromotive force (EMF). Emf is a potential difference between two points (cathode and anode) of the battery. As a result, the voltage increases as the potential difference increases. Then how is a metal’s respective standard reduction potential calculated? First, you need to make a battery: the target metal as cathode with a standard hydrogen electrode (SHE) acting as anode. And, measure the electromotive force on that battery. This calculation should occur on all metals under the same condition of temperature, pressure, and even standard hydrogen electrode being used. That is why the word “standard” is attached to both the standard reduction potential and the standard hydrogen electrode.
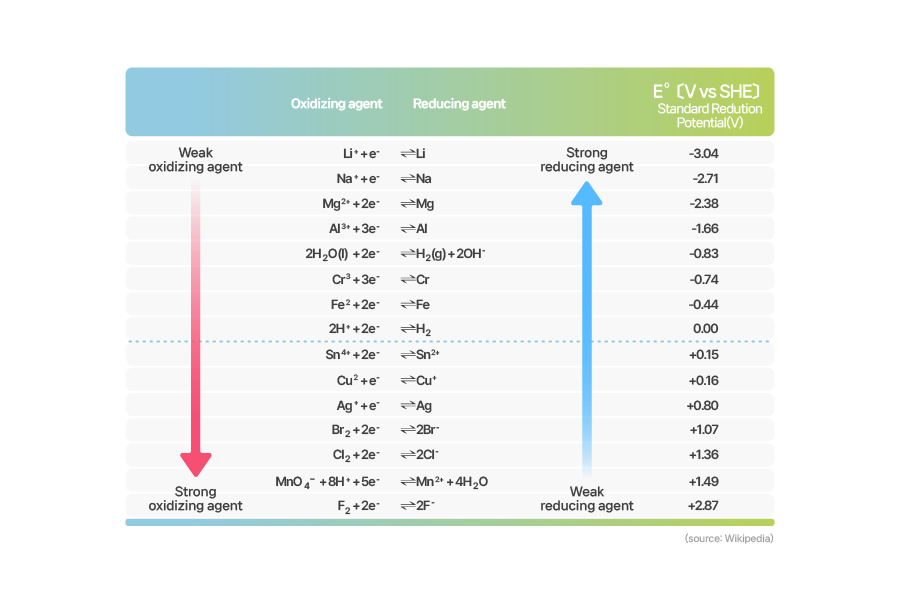
[Standard Reduction Potential]
So that table above is the result of all those calculations on the metals referred in the chart of the ionization energy. You can see there’s a tendency: the higher the ionization energy, the lower the standard reduction potential. A lower standard reduction potential value means that the metal has bigger reactivity.
The standard reduction potential of hydrogen is zero, as you see in the table up there. That is because hydrogen was acting as anode, stopping any reduction-oxidation reactions from happening. This leads to a conclusion that if a metal holds a higher reduction potential value than that of hydrogen, it is more prone to reduction, whereas a lower value indicates a more tendency to oxidation.
You can make a self-running chemical cell if you connect a high reduction potential metal (reduction agent) on the cathode and a low one on the anode. The table of the standard reduction potential also gives us the calculation for theoretical electromotive force of a battery.
